SAM Detects Some Curious Carbon At Gale
A recent paper published in PNAS reported SAM’s discovery of some interesting carbon from samples at Gale crater. Carbon is a necessary element for life as we know it and its isotopes can tell us about ancient processes. Carbon has two stable isotopes: 12C, which makes up the majority of all carbon, and 13C, which represents the remaining carbon. The superscript numbers are based on the number of protons and neutrons in the atomic nucleus, where 12C has 6 protons and 6 neutrons while 13C has 6 protons and 7 neutrons. The relative amounts of 13C and 12C can help reveal chemical and biological processes. SAM can analyze carbon and its isotopes from samples to help piece together the history of carbon on Mars.
SAM analyzed two dozen solid samples collected from Mars and found some intriguing isotopic signatures. These samples were heated to a high temperature (~850 ºC), which resulted in the decomposition of sample components and the corresponding release of gases such as carbon dioxide (CO2), water (H2O), and methane (CH4). The gases were sent to the tunable laser spectrometer, which can determine their abundances and isotopes. The team found that 10 of these 24 samples from 6 unique locations released methane whose carbon had much less 13C than 12C compared to an Earth standard (in other words, they were depleted in 13C relative to the standard). These highly depleted values were all at least 70 parts per thousand (‰) less than the standard (i.e., < -70‰). For comparison, the CO2 in the modern Martian atmosphere has an overabundance of 13C compared to the Earth standard by +46‰. Interestingly, in addition to the highly depleted carbon isotopes, the team found broad associations with evidence for reduced sulfur (like the mineral pyrite, a.k.a. fool’s gold), isotopes of sulfur that were also low compared to a standard, and sample sites that may have been connected by an ancient surface. The anomalous carbon isotope values are shown below in Figure 1.

Figure 1. From House et al. (2022) showing sample sites with strongly depleted 13C values.
What could be causing such highly depleted values? The team put forward three hypotheses (summarized in Figure 2, below) that all fit the data, but none of them can be accepted or rejected without additional data and analysis. Let’s dig into each of these hypotheses a bit.
- Biologically-produced methane reacted with ultraviolet (UV) radiation in the atmosphere and deposited as organic molecules onto the paleosurface. Life on Earth has a preference for the lighter isotope of carbon, 12C. Methane can be produced by some microbes on Earth and tends to be depleted in 13C. If Martian microbes were present and produced methane, it could have interacted with ultraviolet radiation to produce organic molecules (carbon-containing compounds) that were deposited. Similarly, microbes on Earth can have metabolisms that result in depleted sulfur isotopes and reduced sulfur. While microbes could, in principle, explain the carbon and sulfur isotope depletions, other evidence is needed before accepting a biological explanation. Such an explanation would have required a complex network of microorganisms and there has been no evidence that such communities were present on Mars, unlike on Earth where such evidence is abundant in the rock record.
- Atmospheric CO2 reacted with ultraviolet radiation and sulfur dioxide (SO2) to form isotopically depleted organics. Some studies have shown that ultraviolet light can form carbon monoxide (CO) from CO2 with carbon depleted in 13C concentrating in the CO in certain conditions. If this happened on Mars, CO may have undergone further reactions to form more complex organic molecules (including some containing sulfur) that were deposited onto the proposed ancient surface, perhaps shortly after a volcanic eruption. It is not yet clear, however, that ultraviolet radiation in the Martian atmosphere would produce 13C-depleted CO. While this explanation is plausible, more research about the isotopic effects of radiation under Martian conditions needs to be conducted.
- Mars passed through a giant molecular cloud (GMC), which may have deposited isotopically depleted dust onto an ancient Martian surface. Our solar system passes through a giant molecular cloud approximately every 100 million years. The dust in these clouds can contain carbon that is highly depleted in 13C (down to -260‰) that could explain the values seen in some of the samples on Mars. This dust may have caused a cooling event that allowed for the formation of glaciers. The dust then could have been concentrated on glaciers that later melted to deposit the carbon on an ancient surface. This hypothesis, while it explains the carbon isotope data, may be too reliant on a rare event, and there is not currently strong evidence for glaciers associated with the proposed ancient surface. More research is needed to definitively rule this hypothesis in or out.
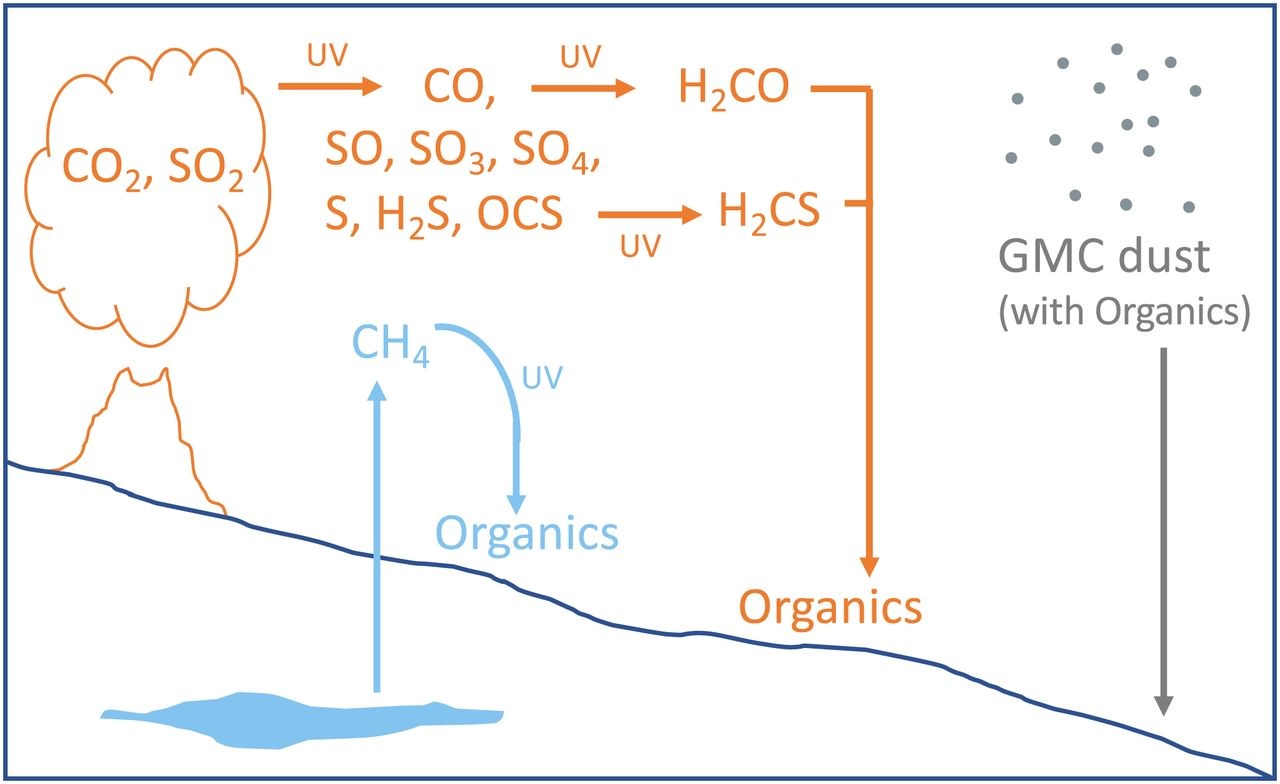
Figure 2. From House et al. (2022) summarizing the three hypotheses above. In blue: methane produced by microbes is released to the atmosphere, reacts with UV light, and falls to the surface as organic molecules. In orange: gases from a volcanic eruption go through a series of reactions with UV light to eventually deposit organics to the surface. In gray: Mars passes through a giant molecular cloud that deposits dust onto the Martian surface.
All of these hypotheses could reasonably fit the data; however, more work needs to be done before we can land on an explanation. The rover will have the opportunity to return to the proposed paleosurface and we hope to look for more clues!